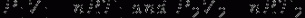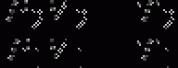Tire Pressure
An automobile tire is filled to a gauge pressure of 167 kPa when its temperature is 13°C. After the car has been driven at high speeds, the tire temperature has increased to 48°C. Calculate the gauge pressure if the tire expands such that the volume increases by 7 percent. (Note: Pgauge = Pabs - 1 atmosphere.)
180 kPa
180 kPa
Approach
We can use the ideal gas law to find the gauge pressure at 48°C.
SolutionThe initial pressure of the tire is 167 + 101.3 = 268.3 kPa. Since no air leaks out we have equation 1
Dividing the second equation by the first we have equation 2 In this case, V2 = (100+7)/100 V1 = 1.07 V1. Everything else remains unchanged. So equation 3 where the temperatures are in in Kelvin. Solving gives P2 = 281.4 kPa. Or the gauge pressure is 281.4 - 101.3 = 180 kPa. |