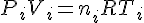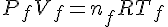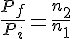Constant Volume
The pressure in a helium gas cylinder is initially 27.7 atmospheres. After many balloons have been blown up, the pressure has decreased to 5.05 atm. What fraction of the original gas remains in the cylinder?
0.182
0.182
Approach
The quantity that measures the amount of gas is the number of moles. To find the fraction of original gas that remains, we need to find nf/ni, both of which can be obtained using the ideal gas law.